Table of Contents Show
Skin, the outermost protective sheath of our body, remains most exposed to the environment. As a result, several bacteria, fungus, and other infectious agents find it their most favorable target.
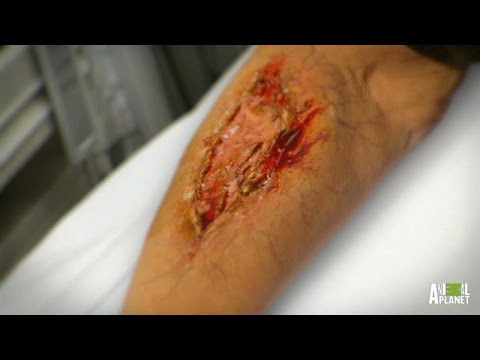
Flesh eating disease or necrotizing fasciitis is one of the most dreadful skin infections in which the skin and the superficial layers beneath it gets affected. It is an infrequent infection in which out of every 4 infected people, one gets killed.
History of Necrotizing Fasciitis (Flesh Eating Disease)
In the 5th century BCE, Hippocrates first described the necrotizing soft tissue infection in which erysipelas spreads all over the body. Bones, flesh, tendons would fall from the body and even cause death.
British surgeon Leonard Gillespie and British physicians Gilbert Blaine and Thomas Trotter in the 18th century gave descriptions for this kind of necrotizing soft tissue infections.
Various names were assigned to this disease, such as phagedenic ulcer, gangrenous phagedena, gangrenous ulcer, malignant ulcer, putrid ulcer, galloping gangrene, hospital gangrene.
In 1883, Dr. Jean Alfred Fournier described the necrotizing of the perineum and scrotum, which came to be known as Fournier Gangrene. The term necrotizing fasciitis was coined by Dr. Wilson in the year 1952.
Target areas of necrotizing fasciitis
Although termed as a skin infection, necrotizing fasciitis can also spread to layers below the skin and is also known as soft tissue infection.
It is a bacterial infection spreading from the skin’s surface to the connective tissue beneath it called the superficial fascia.
The infection spreads very fast all over the body and turns into a fatal one causing tissue death.
Signs and symptoms of necrotizing fasciitis
The initial symptoms are often quite lenient and have similarities to other skin diseases making it quite delayed to be diagnosed. The symptoms gradually change at various stages of the development of the disease.
Early symptoms include-
- The skin becomes red, warm and swelling gradually spreads from one place to another. This is often followed by severe pain.
- The swollen skin and the soft tissue gradually get hardened.
- Sometimes fever also becomes a part of the initial symptoms.
Later symptoms include-
As the severity of the infection intensifies with time, it picks up some recognizable and unique critical symptoms of necrotizing fasciitis. These include-
- Formation of bullae and bleeding into the skin are the symptoms just before the severity of the infection reaches a maximum level.
- Due to thrombosis of the blood vessels, the skin color gradually changes from red to purple and then finally to black.
- There is often gas formation in the tissues, along with reduced sensitivity, and the necrotizing infection spreads, causing too much tissue damage.
- Blisters and ulcers also start appearing on the skin.
- Sometimes even pus oozes out of the infected area.
- It may also be accompanied by dizziness, fatigue, diarrhea, and other serious complications that may indicate that one is developing necrotizing fasciitis.
The flesh eating bacteria: the causative agent
Necrotizing fasciitis is a bacterial infection that can be categorized into four types, namely-
- Type 1 infection
- Type 2 infection
- Type 3 infection
- Type 4 infection
Type 1 infection
- This is the most common type of infection that accounts for nearly 70%-80% of all global cases. This is caused by a mixture of bacterial types, which includes Gram-positive cocci and gram-negative rods, as well as by anaerobes.
- Necrotizing fasciitis flesh-eating Gram-positive bacteria include Staphylococcus aureus, Streptococcus pyogenes, and enterococci.
- Gram negative bacteria include Escherichia coli, Pseudomonas aeruginosa etc.
- Anaerobes like Bacteroides and Clostridium species also cause this kind of skin infection.
- This is mainly found in medical comorbidities like diabetes mellitus, obesity, and immunodeficiency.
- The Clostridium species produces two types of toxin, namely alpha and theta toxin, both of which facilitate the blockage of red blood cells inside the blood vessels and prevent the functioning of the white blood cells and thus inhibiting their phagocytic function and may even suppress the heart functioning.

Type 2 infection
- This is mainly found in young adults with a history of injury.
- The main culprit behind this infection is Streptococcus pyogenes which produce M protein which acts as a superantigen, which in turn causes a massive immune attack manifesting as streptococcal toxic shock syndrome leading to multiple organ failure.
- This accounts for 20%-30% of all infections.
Type 3 infection
- It is quite a rare disease.
- It is caused by Vibrio vulnificus, which is found in salty water. It generally paves its way through any minor wounds and cuts.
- The signs and symptoms are generally the same as type 2, but there might be some more visible skin color and texture changes.
Type 4 infection
- According to some scientists, it may be due to some fungal infections.
Vulnerable groups-
Although necrotizing fasciitis (flesh eating disease) can attack anyone, some are more susceptible to this disease. People with other serious health issues are more likely to get this disease.
If someone has weakened immunity, they can likely develop the symptoms. The following group is likely to be the primary target site of the bacteria-
- Individuals addicted to alcohol and other drugs are more likely to get these diseases.
- Those who have a cancer history or suffering from cancer can also be prey to this bacterial infection.
- Those who have developed chickenpox in childhood.
- For those who have diabetes and cirrhosis and the body’s immune system gradually gets weakened.
- Those suffering from heart disease and chronic kidney disease.
- Those having lung disease including tuberculosis, peripheral vascular disease.
- The use of steroids also increases the chance of the disease.
- Those who have surgical wounds are also more likely to develop this kind of skin infection.
Diagnosis of Flesh Eating Disease-
The sooner Necrotizing Fasciitis is detected, the better it is. Early diagnosis is the best treatment. The more the diagnosis gets delayed, complications rise, making it a life-threatening one.
As the initial symptoms are quite mild, they are often misdiagnosed as mild skin infections, and this delay ultimately puts the patient in a life-risking condition.
There are various ways to diagnose as well as to confirm the disease; some of the most common are-
The gold standard for diagnosis-
The confirmatory test for Necrotizing Fasciitis is an invasive surgical procedure. In a doubtful case, a small incision is made into the affected tissue, and if a finger easily separates the tissue along the fascial plane, the diagnosis is confirmed.
It is an exploratory surgery where it is common foul-smelling discharge, necrosis, and loss of the normal resistance of the fascia to finger dissection.
Medical Imaging-
Although this can not confirm the presence of Necrotizing fasciitis, it can give hints about its presence. This is done through radiographic techniques like X-rays, Computed Tomography(CT scans), Magnetic Resonance Imagining(MRI)
- X -rays are not the preferable choice for detecting necrotizing fasciitis as this only captures the air-filled cavities under the skin called the subcutaneous emphysema. This is not a characteristic of Necrotizing fasciitis as it is present only in a small proportion of these patients.
- CT scans are readily available and are one of the best methods to identify this soft tissue infection. Through CT scans, doctors can get a detailed image of inflammatory changes, such as accumulation of fluid called edema, thickening, or collection of pus called abscesses in the fascia, in addition to gas formation. CT scan can detect nearly about almost 80% of all cases.
- Magnetic Resonance Imagining is a good way to get an almost detailed report of Necrotizing Fasciitis. Through this procedure, fascial thickening of the soft tissue can be diagnosed.

Laboratory methods-
Through fluid body analysis, one can also come to an approximate conclusion about the presence of this disease.
If an individual has a white blood cell count of more than 15,400cells /mm3and a sodium level lower than 135mmol/L, there may be the chances that they are a patient of Necrotizing Fasciitis.
Although there are several scoring methods, a scoring system developed by Wong and colleagues in 2004 is the most commonly used. The scoring method stands as-
- CRP (mg/L)
≥150: 4 points
- WBC count /×103/mm3
<15: 0 points
15–25: 1 point
>25: 2 points
- Hemoglobin (g/dl)
>13.5: 0 points
11–13.5: 1 point
<11: 2 points
- Sodium (mmol/l)
<135: 2 points
- Creatinine (umol/l)
>141: 2 points
- Glucose (mmol/l)
>10: 1 point
A score of 6 or more is considered a severe case of Necrotizing Fasciitis. Although it has been found that some Necrotizing Fasciitis patients have scored less than 6, this gives the preliminary finding for Necrotizing Fasciitis.

All these are the most common ways to diagnose Necrotizing Fasciitis.
Treatment for Necrotizing Fasciitis-
Depending on the severity of Necrotizing Fasciitis in the infected person, the doctors can decide which is the best way or option to treat this disease. Following pathways are generally chosen to treat this infectious disease-
- surgical debridement,
- antibiotic therapy,
- hyperbaric oxygen therapy,
- IV immune globulin (IVIg) therapy.
Surgical debridement-
- The best option is debridement of the dead tissue at the infected site as soon as the disease is diagnosed. This shall save the rest of the healthy tissue and blood vessels and prevent necrotizing soft tissue infections. The debridement is often carried in areas more than the area of induration. Sometimes the cellulitic soft tissues are kept away from debridement for later skin coverage of the wound.
- In most cases, it is found that a single surgery is not sufficient for the removal of all the infected tissue. Often the entire muscle or amputation of limbs may be needed. It may require multiple surgeries in a time gap of 12 to 36 hours to fight infection.
- After surgery, the proper wound dressing is a must so that the cartilage, bones, and other layers remain in place and not get further infected.
Antibiotic Therapy-
Although antibiotics can not reach all the infected tissues, they can still inhibit sepsis and further spread of the infection. It is generally given as a part of follow-up after the surgery.
As antibiotics fail to reach all the infected tissues, surgery remains the first choice. Although there is no established duration for antibiotic therapy, it can still be continued as long as no more surgical debridement is required or there is no inflammation. The two most commonly used antibiotic combinations are-
1. Vancomycin, Clindamycin, and Piperacillin/ tazobactam
2. Linezolid and Piperacillin/tazobactam
Hyperbaric Oxygen Therapy-
This procedure has not been implemented on a large scale and remains controversial if one can be cured with this procedure. Clinical trials are going in both humans and animals. In due course of time, the efficiency of this method will be known.
Various species of Clostridium1, like Clostridium perfringens infection and exotoxin production, can be inhibited in hyperbaric conditions. But it is found that the Necrotizing Fasciitis caused by Clostridium is quite less common and hence the efficiency of this method is a matter of thought.
Immune globulin (IVIg) therapy-
Immunoglobins are antibodies produced in our body and found in our body fluids. The antibodies attach to foreign agents like fungus and bacteria and help the immune system destroy them. They play a vital role in the defense mechanism of the immune system.
Although IVIg therapy is not an FDA2-approved one, it has been found to minimize the effect of exotoxin produced by Streptococci sp. and Staphylococci sp.
There has been a little human trial on this method, and thus more researches are being conducted to develop this method in a safe way for humans.
How to prevent Necrotizing Fasciitis and other skin infections-
Cleanliness and proper wound care is the best method to prevent Necrotizing Fasciitis.
The ways to get rid of Necrotizing Fasciitis and other skin infections include-
- One should clean cuts and open wounds with soap and water.
- After that, covering an open wound with a clean bandage is a must.
- One should seek a doctor’s consultation if one has a severe or deep wound, for example, a gunshot.
- One should avoid swimming or using a hot tub if one suffers from skin infections.
- One should also care for fungal infections.

Related disorders-
Necrotizing Soft Tissue Infection (NSTI)-
Necrotizing Fasciitis is a part of Necrotizing Soft Tissue Infection (NSTI). NSTI includes all those infections that cause harm to soft tissues.
This terminology is used by doctors since all NSTI 3require a similar approach to diagnosis and remedy irrespective of the location of infection on the body.
Some examples of NSTI are necrotizing adiposity or necrotizing cellulitis, necrotizing fasciitis, necrotizing myositis.
Toxic Shock Syndrome (TSS)-
When a patient is infected with a bacteria and succumbs to the exo-toxins4 and endo-toxins released by the bacteria, there may be a chance of getting toxic shock syndrome. The ways to manage the syndrome includes-
- antibiotics,
- fluid management
- vasopressors.
Typical symptoms for TSS includes-
- high fever,
- low blood pressure,
- rashes, vomiting,
- diarrhea,
- kidney failure,
- low platelet counts,
- confusion.
Mucormycosis-
The Mucorales fungi cause Mucormycosis. It is found in bread or fruits, and it generally does not affect healthy individuals, but those with immunodeficiency can fall prey to this fungus.
Infection caused by fungi does not respond to antibiotics. The best remedy is early diagnosis, followed by proper treatment and anti-fungal drugs, reducing severity.
Insect Bites-
Insect bites can also cause Necrotizing Fasciitis. These bites are not generally fatal but can cause pain, redness, and swelling. Proper wound care and the use of antibiotics are the best treatment options.
Necrotizing Fasciitis- Contagious or not
Necrotizing Fasciitis occurs randomly. There can be sudden onset of this soft tissue infection in any individual. Reports are not found that Necrotizing Fasciitis can be contagious and spread from one individual to another.
So doctors generally do not prescribe any preventive antibiotics 5for those in close contact with such patients.
Last Words:
The infection of Necrotizing Fasciitis is a challenging task for both the patient and doctor. Early diagnosis can prevent amputation of body parts and vital organs. To avoid any infection or disease, good health is the best solution.
The healthier and more clean lifestyle we lead, the less the chances of falling prey to diseases.
Physicians and scientists all over the globe are trying to find better ways to curb the severity of the infection. Although immunodeficient people are more susceptible to this kind of skin infection, we all must care for it.
As it turns life-threatening in later stages, we must seek medical consultation as early as possible. The early steps of today can save a life in the future.
Click here to read more.

- Guo, Pingting, et al. “Clostridium species as probiotics: potentials and challenges.” Journal of animal science and biotechnology 11 (2020): 1-10. ↩︎
- Darrow, Jonathan J., Jerry Avorn, and Aaron S. Kesselheim. “FDA approval and regulation of pharmaceuticals, 1983-2018.” Jama 323.2 (2020): 164-176. ↩︎
- Pelletier, Jessica, et al. “Necrotizing soft tissue infections (NSTI): Pearls and pitfalls for the emergency clinician.” The Journal of Emergency Medicine 62.4 (2022): 480-491. ↩︎
- Derakhshan, S., A. Rezaee, and Mohammadi Sh. “Relationship between type III secretion toxins, biofilm formation, and antibiotic resistance in clinical Pseudomonas aeruginosa isolates.” Инфекция и иммунитет 11.6 (2021): 1075-1082. ↩︎
- Butler, Mark S., and David L. Paterson. “Antibiotics in the clinical pipeline in October 2019.” The Journal of antibiotics 73.6 (2020): 329-364. ↩︎
Last Updated on by Sathi Chakraborty, MSc Biology